 When doing a serial dilution, the dilution factor for either each test tube or the total dilution factor for the whole series may be approximated.Sequential two-fold as well as ten-fold dilutions are frequently used in the lab to titer antibodies or manufacture diluted analytes.On the other hand, a sample that is less contaminated may only need a little dilution factor. For example, if a water sample is taken from a location with significant levels of contamination, the dilution factor is increased. Based on the anticipated quantity of cells or other organisms present in a sample, the degree of dilution is determined.Later, employing a modest calculated amount of each dilution, a sequence of pour or spread plates are made.A consistent quantity of sterile diluent, such as distilled water or 0.9 percent saline, is used to dilute a sample during the serial dilution procedure.By using dilution to generate incubated culture plates containing a countable number of colonies, it is possible to calculate the quantity of microorganisms contained in a sample (between 30 and 100).Serial dilutions are frequently used to dilute a solution instead of pipetting extremely small volumes (1–10 l).To make it simpler to determine the concentration of the cells in the original solution by determining the overall series’ total dilution, serial dilution reduces the density of cells at each phase.The serial dilution approach seeks to determine the concentration (number of organisms, bacteria, viruses, or colonies) of an unknown sample by calculating the number of colonies that have been produced from subsequent dilutions of the sample. 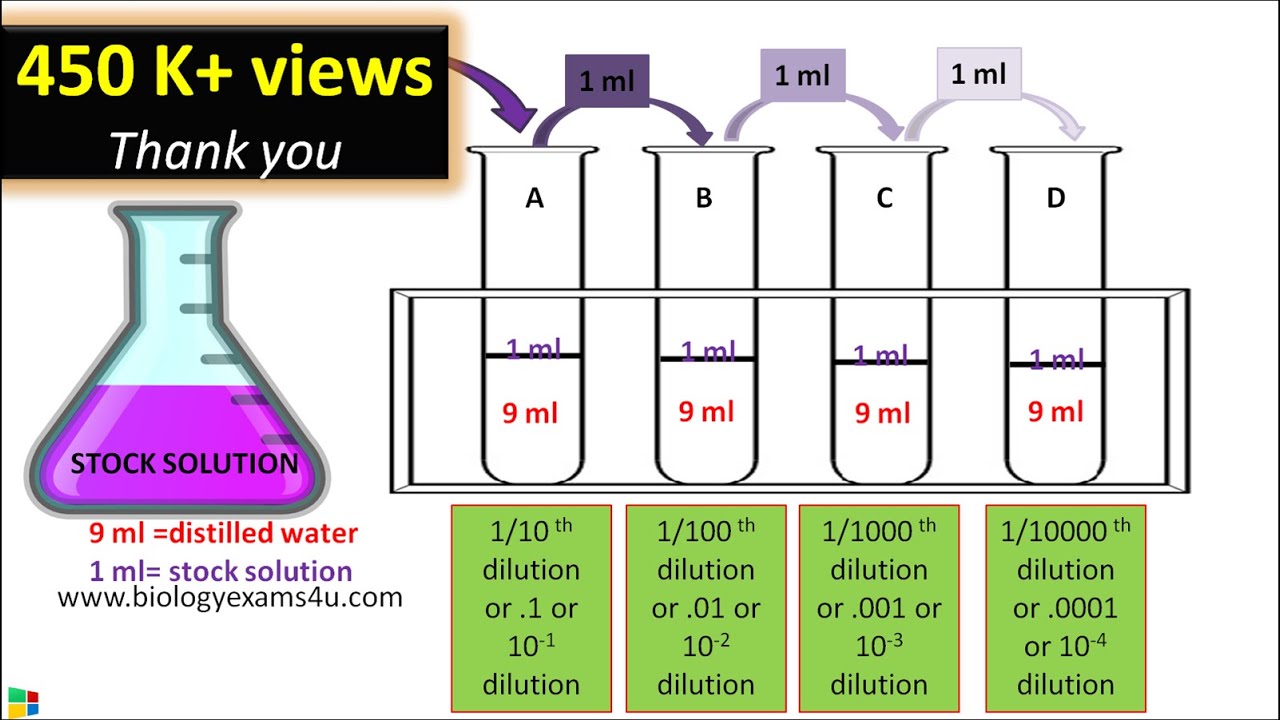
Like the title indicates, a sequence of consecutive dilutions may be used to transform a thick solution into a concentration that is more usable. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
